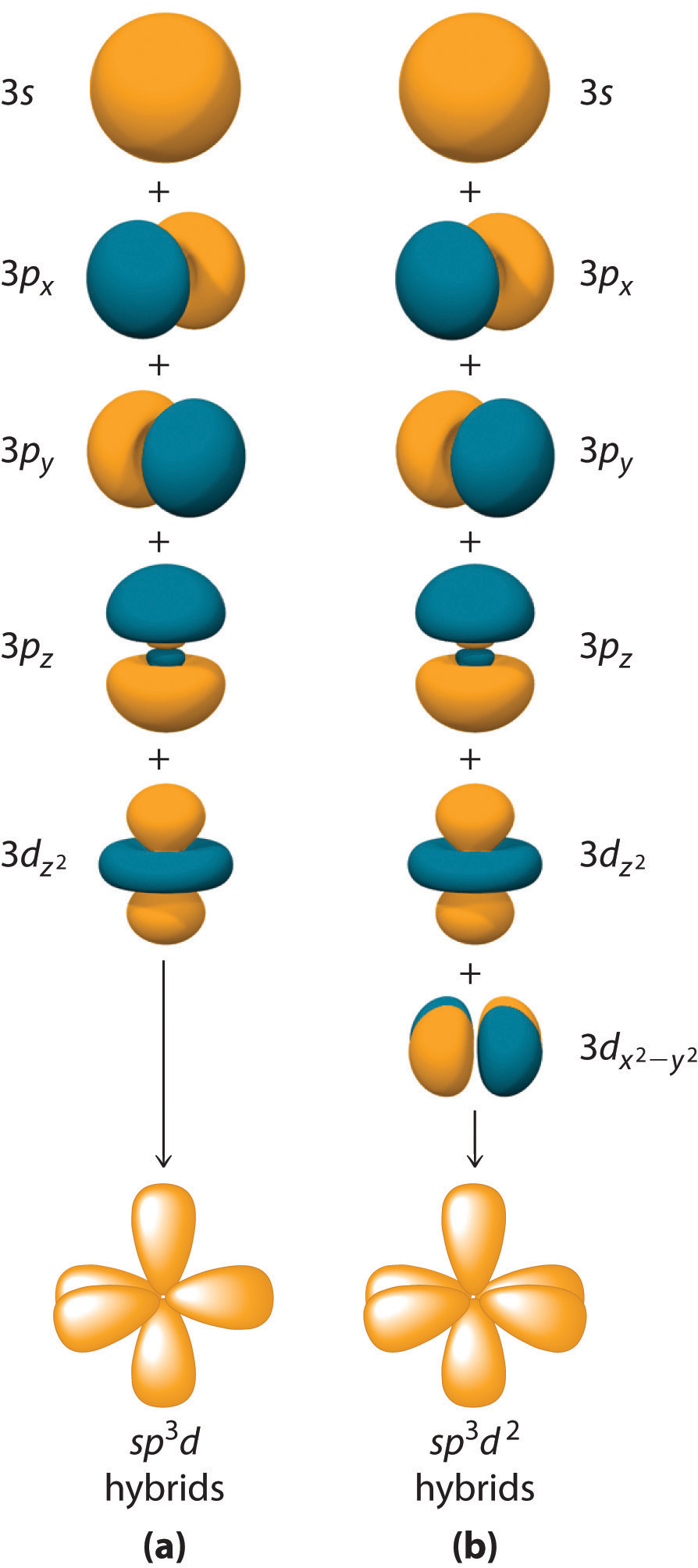
I think that the explanation in terms of molecular orbitals and in terms of atomic orbitals are unique and each contribute in separate ways, so there may be merits for leaving it there. I feel like it is explained a little better with the discussion of methane, though everything prior to where the stress exists that orbitals are models could be considered unnecessary. But you're right, the molecular orbital page certainly does do much of the pre-hybrid explanation work. I realized it would be fairly redundant, though I don't believe a page exists for any hybrids except sp², which right below is indicated as needing work as well. rmbh 07:34, (UTC) I was thinking about it over the weekend, and I was considering expanding on the bottom section and discussing in more detail the various forms of hybridization. The specialized hybridized orbital pages should probably be incorporated here, instead of the bulk of it, which is better explained in the molecular orbital page. Oh wow! The molecular orbital page builds all the way to hybridization, and it doesn't link to this page! There's been a lot of duplicated effort 's really the molecular geometry concepts that are unique to this article. So I think this could use more work (yes, I'll do some!) rmbh 07:08, (UTC) This would improve the understanding that hybridized orbitals are the result of multiple basis states existing in superposition, with a significant energetic stabilization due to resonance in hybridized orbitals compared to atomic ones. Then, the hybridized orbitals can be introduced as a mixing of the atomic orbitals, as a linear combination of basis (linear algebra)states. I'm going to see if something like that exists. I think another article shoud introduce the theory of atomic orbitals, in terms of the solutions to the Schrodinger equation for electrons. There's perhaps just two much writing that's introductory, and it makes it a bigger read than necessary. I think it could have more cross-referencing for the introduction, and a more focused body. Preceding unsigned comment added by EagleFalconn ( talk I've got it on watch, so any sugestions along the way are welcome. Over the next several days/several edits I'm going to try and overhaul this article and make it more comprehensible. 34 hybridization and electronic configuration.33 Draft new section on hybridization of heavier p block elements.31 Two sources are often better than one.30 An inconsistency and a question or two.

28 orbital hybridisation of hypervalent molecules.25 Hypervalent molecules expand their octets or dodectets.20 Controversy regarding d-orbital participation.
This article has been rated as C-Class on the project's quality scale. Chemistry Wikipedia:WikiProject Chemistry Template:WikiProject Chemistry Chemistry articles If you would like to participate, please visit the project page, where you can join the discussion and see a list of open tasks. This article is within the scope of WikiProject Chemistry, a collaborative effort to improve the coverage of chemistry on Wikipedia. According to the relevant style guide, this should not be changed without broad consensus. This article is written in British English, which has its own spelling conventions ( colour, travelled, centre, realise, defence, artefact), and some terms that are used in it may be different or absent from other varieties of English.
